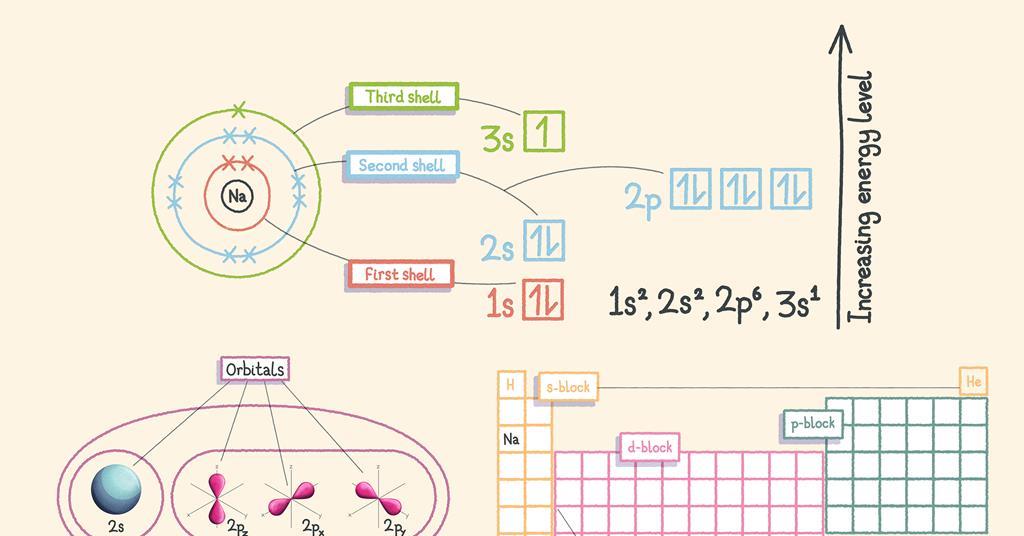
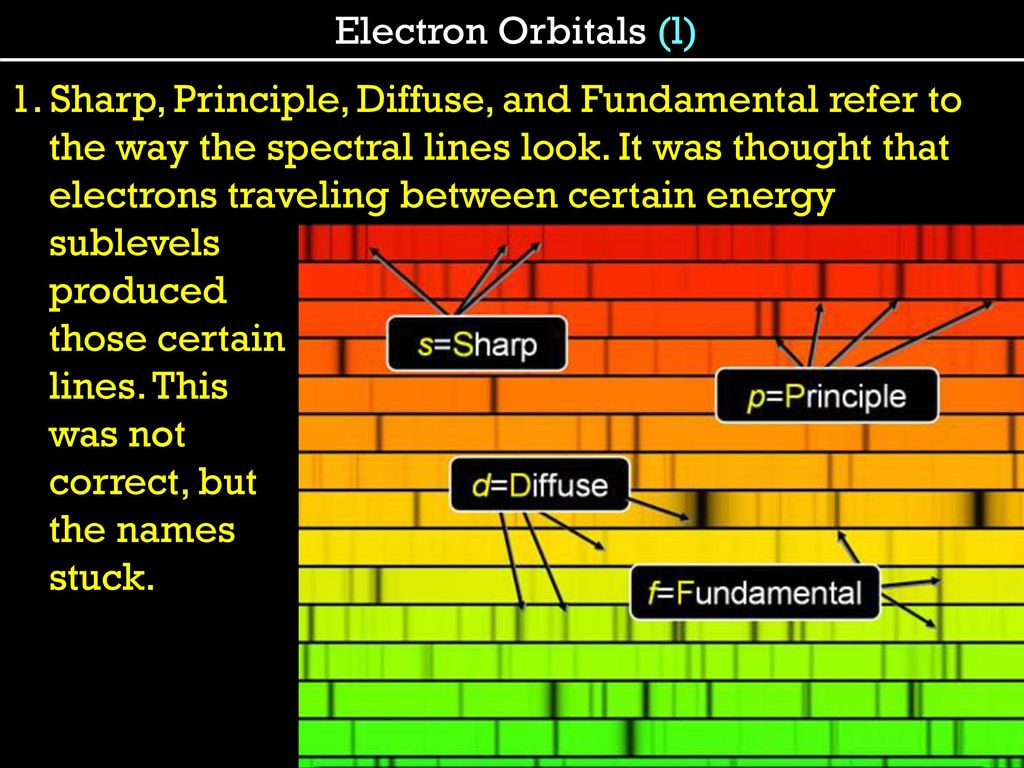
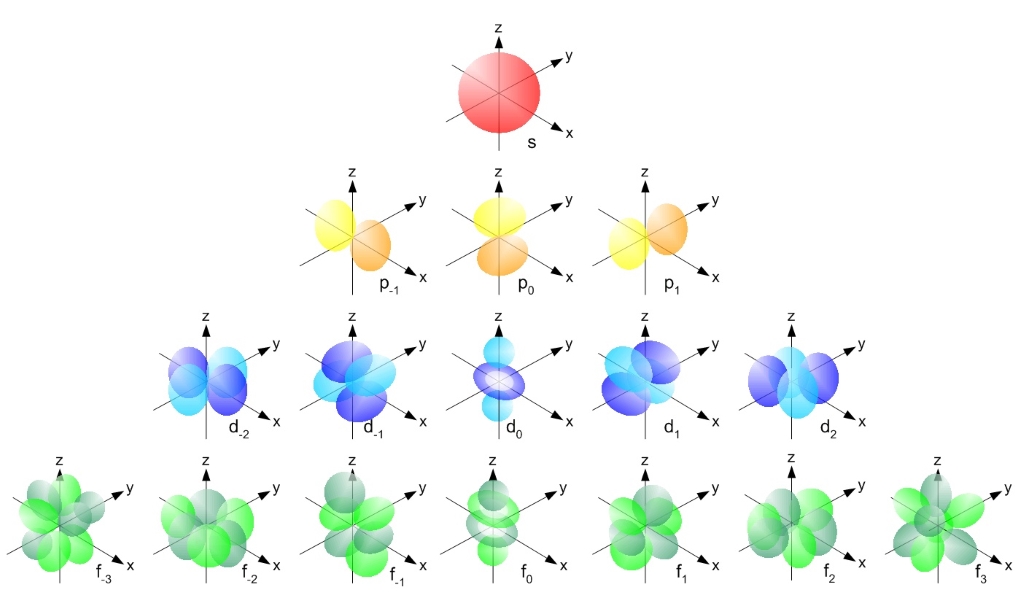
Eigenbros on X: "Ever wondered why atomic orbitals have the letters they do? They were named corresponding to their spectral lines in alkali metals #Physics s - sharp p - principal d -
The subsidiary/azimuthal quantum number is s, p, d, f, g, h and so on. where the s, p, d, and f stand for sharp, principal, diffuse and fundamental, respectively. What is the
What does it mean when orbitals are called sharp (s orbital); principle (p orbital); diffuse (d orbital) and fundamental (f orbital)? - Quora